On June 10, 2019, Texas Governor Greg Abbott signed HB 1325, legislation pertaining to hemp growth and consumable hemp products, into law in the state of Texas. To conform with Texas Health and Safety Code, Chapter 443, as amended by HB 1325, Texas Department of State Health Services (“DSHS”) has published its proposed rules to govern the Texas consumable hemp program (the “DSHS Proposed Rules”) in the Texas Register. Under the DSHS Proposed Rules, a “consumable hemp product” is defined as
Any product processed or manufactured for consumption that contains hemp, including food, a drug, a device, and a cosmetic, as those terms are defined by Texas Health and Safety Code, §431.002, but does not include any consumable hemp product containing a hemp seed, or hemp seed-derived ingredient being used in a manner that has been generally recognized as safe by the [United States Food and Drug Administration]. Title 25 TAC § 300.101(8).
This definition of consumable hemp products encompasses all foods, drugs, devices, and cosmetics containing properly-sourced hemp. Under HB 1325, a consumable hemp product derived from properly-sourced hemp that contains no more than .3% THC on a dry-weight basis is a legal good in Texas, as long as it is sourced and produced in accordance with HB 1325 and the relevant TDA and DSHS regulations. Additionally, HB 1325 requires DSHS to oversee and regulate the manufacture, processing, distribution, and sale of consumable hemp products.
DSHS Proposed Rules were published on Friday, May 8, 2020, and will be open for public comment for 31 days. If you are dissatisfied with any provision of the DSHS Proposed Rules or desire to comment on any of the draft rules, you may submit comment directly to DSHS by email to DSHSHempProgram@dshs.texas.gov. When emailing comments, please indicate “Comments on Proposed Rule 19R074 Hemp Program” in the subject line. Written comments on the proposal may also be submitted to Rod Moline, Ph.D., R.S., Section Director, Mail Code 1987, Texas Department of State Health Services, P.O. Box 149347, Austin, Texas 78714-9347.
In Parts I through III of this Series, we will discuss the following: general provisions, ban on smokables, and inspections pertaining to the manufacture and retail of consumable hemp products; application and license requirements for consumable hemp product manufacturers, processors, and distributors; and methods and regulations for the sampling and testing of consumable hemp products. In Parts IV through VI of this Series, we will analyze the following: the application and registration requirements for consumable hemp product retailers; the requirements imposed for the retail sale of consumable hemp products; and violations and enforcement protocols under the DSHS Proposed Rules.
Part I: General Provisions, Ban on Smokables, and Inspections
General Provisions
Before getting into the complexities of the DSHS Proposed Rules, it is important to note the various other laws that must be complied with. In addition to complying with HB 1325, the DSHS Proposed Rules, and federal law, hemp distributors, manufacturers, processors, and retailers must comply with the following statutes (if applicable to their particular industry):
- (1) Chapter 217, Subchapter C of this title (relating to Rules for the Manufacture of Frozen Desserts);
- (2) Chapter 229, Subchapter D of this title (relating to Regulation of Cosmetics);
- (3) Chapter 229, Subchapter F of this title (relating to Production, Processing, and Distribution of Bottled and Vended Drinking Water);
- (4) Chapter 229, Subchapter G of this title (relating to Manufacture, Storage, and Distribution of Ice Sold for Human Consumption, Including Ice Produced at Point of Use);
- (5) Chapter 229, Subchapter L of this title (relating to Licensure of Food Manufacturers, Food Wholesalers, and Warehouse Operators);
- (6) Chapter 229, Subchapter N of this title (relating to Current Good Manufacturing Practice and Good Warehousing Practice In Manufacturing, Packing, Or Holding Human Food);
- (7) Chapter 229, Subchapter W of this title (relating to Licensing of Wholesale Distributors of Prescription Drugs–Including Good Manufacturing Practices);
- (8) Chapter 229, Subchapter X of this title (relating to Licensing of Device Distributors and Manufacturers); and
- (9) Chapter 229, Subchapter GG of this title (relating to Sanitary Transportation of Human Foods). Title 25 TAC § 300.102.
If one of these statutes applies to your industry, compliance is mandatory.
Ban on Smokables
Another key general provision under the DSHS Proposed Rules is the provision banning the “smoking” of consumable hemp products. DSHS defines “smoking” as “[b]urning or igniting a consumable hemp product and inhaling the resultant smoke, vapor, or aerosol.” Id. at § 300.101(35). Under the DSHS Proposed Rules, the distribution, processing, manufacture, and retail sale of consumable hemp products for smoking is illegal. See id. at § 300.104. Of note, HB 1325 did not ban the retail sale of smokable hemp. DSHS is imposing a new requirement above the statutory restriction on manufacturing of smokable hemp products. This outright ban of smokable hemp products, such as vapes, will negatively impact Texas growers and retailers who were expecting to generate significant revenue from smokable products. Texas consumers can still purchase these products out-of-state, but this proposed rule will be a huge detriment to in-state businesses seeking to profit from smokable hemp products. Hemp distributors, manufacturers, processors, retailers, and others in the hemp industry desiring modification or removal of the retail ban on smokable hemp should submit comment to DSHS during the 31-day window open for public comment.
Inspections
Employees authorized by DSHS are to conduct, with reasonable promptness, inspections of facilities where consumable hemp products are distributed, held, manufactured, packed, processed, or sold into commerce for the primary purpose of determining whether the consumable hemp products are adulterated, misbranded, or otherwise sold in violation of this chapter or Chapter 431 (Texas Food, Drug, and Cosmetic Act) and 443 (Manufacture, Distribution, and Sale of Consumable Hemp Products) of the Texas Health and Safety Code. See id. at § 300.103(b).
Inspections of facilities where prescription drugs or restricted devices, as well as consumable hemp products, are distributed, held, manufactured, packed, processed, or sold into commerce shall not extend to pricing data, financial data, sales data other than shipment data, personnel data other than data used for qualification purposes, or research data other than data relating to new consumable hemp products and data subject to reporting and inspection under the Federal Food, Drug, and Cosmetic Act. See id. at § 300.103(c).
Upon presenting proper credentials to the owner or person in charge of the inspected location, authorized employees of DSHS may enter the premises at reasonable times, collect samples, conduct inspections and take photographs to ensure compliance with Chapter 431 and 443 of the Texas Health and Safety Code. See id. at § 300.103(a)(1). These employees also have the option of “enter[ing] a vehicle being used to transport or hold the consumable hemp product in commerce.” Id. at § 300.103(a)(2). DSHS inspectors may “inspect at reasonable times, within reasonable limits, and in a reasonable manner, the facility or vehicle and all equipment, finished and unfinished materials, containers, and labeling of any item and obtain samples necessary for the enforcement of this chapter.” Id. at § 300.103(a)(3). If the inspection parameters seem overly broad or lack clarity, make sure to submit comments and concerns to DSHS during the 31-day public comment window.
Part II: Manufacture, Processing, and Distribution Requirements
Application for License or Renewal
According to DSHS, certain persons are ineligible to apply for a consumable hemp products license. If a person has a felony conviction relating to a controlled substance within 10 years of the application date, or if DSHS later receives information of such a conviction, then that person is restricted from being issued a license, or if they already have a license, then DSHS shall revoke that license. Title 25 TAC § 300.201(c)-(d).
Under the DSHS Proposed Rules, any person seeking to distribute, manufacture, or process consumable hemp and hemp-derived products must annually submit a completed license application, along with the required fees, to DSHS for each location engaged in such activities. Applications must be accompanied by the following information:
- (1) a legal description of each location to include the global positioning system coordinates for the perimeter of each location:
- (A) where the applicant intends to manufacture or process consumable hemp products; and
- (B) where the applicant intends to store consumable hemp products to include the global positioning system coordinates for the perimeter of each location;
- (2) written consent from the applicant or the property owner, if the applicant is not the property owner, for the department, the Department of Public Safety, and any other state or local law enforcement agency, to enter all premises where consumable hemp is manufactured, processed, or delivered, to conduct a physical inspection or to ensure compliance with this chapter; and
- (3) a fingerprint-based criminal background check from each applicant at the applicant’s expense. Id. at § 300.201(b).
In addition to the above-listed application requirements, an application for a consumable hemp products license must also include the following information:
- (1) the name of the license applicant;
- (2) the business name, if different than applicant name;
- (3) the mailing address of the business;
- (4) the street address of the facility;
- (5) the primary business contact telephone number;
- (6) the personal email address of the applicant; and
- (7) the email address of the business, if different than the applicant’s email address. Id. at § 300.201(f).
If a person owns or operates more than one facility, each facility must be licensed separately by including the name and address of each facility on separate application forms. See id. at § 300.201(g).
All applications for consumable hemp products licenses must be submitted electronically, and DSHS has the authority to collect and recover application and renewal application processing costs, in amounts determined by the Texas Online Authority. See id. at § 300.201(h). DSHS will likely create an online application with a specific link, as TDA did for the hemp grow license.
Once a consumable hemp products license is issued, it “should be displayed in an obvious and conspicuous public location within the facility to which the license applies.” Id. at § 300.201(k).
License Terms and Fees
Before a consumable hemp products license holder can distribute, manufacture, or process consumable hemp products, a fee of $250 per facility must be paid. See id. at § 300.202(c)(1). Fees are not prorated under the DSHS Proposed Rules. Additionally, for each facility, a license holder must pay the following fees as necessary:
- (A) a $250.00 fee for amendment to a new license due to a change of ownership of the licensed facility; or
- (B) a $125.00 fee for any amendment during the licensure period due to minor changes, such as change of location, change of name, or change of address. Id. at § 300.202(c)(2).
Applications for amendments are considered to be complete once DSHS has “received, reviewed, and found acceptable the application information and fee required by the subsection (c) of this section.” Id. at § 300.202(d)
Consumable hemp products licenses are valid for one year from the date displayed on the license, and DSHS shall issue and renew a license if the license holder:
- (1) is eligible to obtain a license under §300.201 of this subchapter (relating to Application for License or Renewal);
- (2) submits a license fee to the department;
- (3) does not owe outstanding fees to the department;
- (4) possesses testing results of consumable hemp products before their manufacture, distribution, or sale into commerce, and provides those testing results upon department request; and
- (5) has not been convicted of a felony relating to a controlled substance under federal law or the law of any state in the ten years before the date of renewal of the license. Id. at § 300.202(b).
If a person files a renewal application after the date that their current license expires, they must pay an additional delinquency fee of $100 to DSHS. See id. at § 300.202(c)(4).
For purposes of efficiency, DSHS has implemented specific time periods in which consumable hemp products licenses are to be processed. Under the DSHS Proposed Rules, these licenses shall be processed in accordance with the following time periods:
- (1) the first time period of 45 business days begins on the date the department receives a completed application. If an incomplete application is received, the period ends on the date the facility is issued a written notice that the application is incomplete. The written notice shall be issued within 45 business days after receipt of the incomplete application and describe the specific information or fee that is required before the application is considered complete;
- (2) the second time period of 45 business days begins on the date the department receives a completed application and ends on the date the license is issued or the facility is issued a written notice that the application is being proposed for denial; and
- (3) if the applicant fails to submit the requested information or fee within 135 calendar days after the date the department issued the written notice to the applicant as described in paragraph (1) of this subsection, the application is considered withdrawn. Id. at § 300.202(e).
Essentially, within 45 business days of receiving a consumable hemp products license application, DSHS will either issue a license, provide written notice that the application is being proposed for denial, or provide written notice that the application is incomplete, describing what is required before considering the application to be complete. If the applicant does not respond accordingly within 135 calendar days of DSHS’s issuance of the written notice of incompleteness, then the application will be withdrawn.
If the consumable hemp products license application is not processed in accordance with the above-mentioned time periods, the applicant “has the right to make a written request within 30 business days after the end of the second time period that the department shall reimburse in full the fee paid in that application process.” Id. at § 300.202(f)(1). Basically, an applicant can seek reimbursement of their application fees if the 45-business-day window elapses without DSHS action, but the applicant only has up to 30 business days after that time period to make this written request. If DSHS finds that good cause does not exist for exceeding the time periods, DSHS shall deny the applicant’s reimbursement request and notify the applicant in writing within 30 business days after the decision. See id. at § 300.202(f)(2).
Access to Records
Persons under the DSHS Proposed Rules, or persons already required to keep records under § 519 or § 520(g) of the Federal Food, Drug, and Cosmetic Act, are required to “maintain records on site for immediate inspection, and at the request of the department, provide access to records for review or copying to verify consumable hemp products are being produced in accordance with United States Department of Agriculture . . . or Texas Agriculture Code, Chapter 121.” Id. at § 300.203(a).
Moreover, persons licensed under Chapter 122 of the Texas Agriculture Code (relating to Hemp Cultivation) shall provide to DSHS upon request “the results of tests conducted on samples of hemp or hemp products as evidence that the delta-9 tetrahydrocannabinol content concentration level on a dry weight basis, that, when reported with the accredited laboratory’s measurement of uncertainty, produces a distribution or range that includes a result of 0.3 percent or less delta-9 tetrahydrocannabinol concentration of the hemp or hemp products does not exceed 0.3 percent.” Id. at § 300.203(b). These records must be maintained for at least three years from when they are created. See id. at § 300.203(c).
Part III: Testing of Consumable Hemp Products
Testing and Sampling Requirements
Now that you have read what is required to be issued a consumable hemp products license by DSHS, it is also important to understand the numerous sampling and testing requirements imposed under the DSHS Proposed Rules.
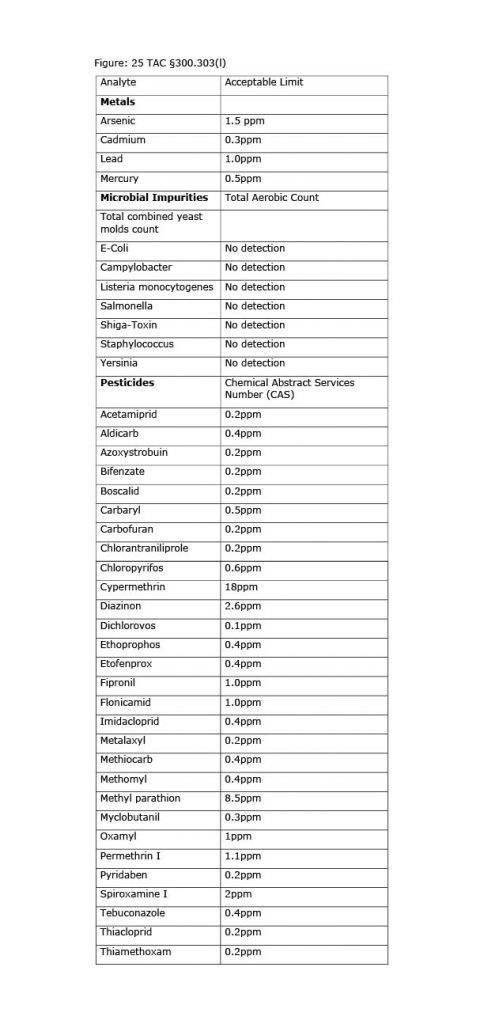
According to the DSHS Proposed Rules, all hemp or hemp derivative products used in the manufacture of consumable hemp products must be tested by an accredited laboratory: “A laboratory accredited in accordance with the International Organization for Standardization ISO/IEC 17025 or a comparable or successor standard.” Title 25 TAC § 300.101(2). These products must be tested to determine the presence and concentration of cannabinoids and of THC, as well as the presence or quantity of harmful pathogens, heavy metals, pesticides, and residual solvents. See id. at § 300.301(a). DSHS may also utilize Table 1, established by the DSHS Proposed Rules, to test the acceptable limits for metals, microbial impurities, and pesticides in raw or finished consumable hemp products. See id. at § 300.303(l).
Additionally, a Certificate of Analysis documenting the accredited laboratory’s testing must:
- (1) be made available to the department upon request in an electronic format before manufacture, processing, or distribution into commerce;
- (2) be in a format that documents presence and content of CBD, and levels of THC; and
- (3) include measurement of uncertainty analysis parameters. Id. at § 300.301(b).
Flexibility in compliance is provided via the “measurement of uncertainty,” which acts as a margin of error in evaluating the accuracy of test results.
DSHS requires that persons may not sell, offer to sell, distribute, possess, or transport consumable hemp products in Texas, including CBD oil, if those products contain any material derived or extracted from the hemp plant, other than from hemp produced in accordance with the United States Department of Agriculture’s (the “USDA’s”) Hemp Production Plan (7 United States Code (U.S.C.) Chapter 38, Subchapter VII), unless:
- (1) a representative sample of the oil has been tested by an accredited laboratory and found to have a delta-9 tetrahydrocannabinol content concentration level on a dry weight basis, that, when reported with the accredited laboratory’s measurement of uncertainty, produces a distribution or range that includes a result of 0.3 percent or less [the ‘acceptable hemp THC level’]; and
- (2) testing results are provided to the department upon request. Id. at § 300.302(b).
It is important to note that a consumable hemp product is not required to undergo the above-mentioned testing requirements if each hemp-derived ingredient of the product has been properly tested, contains the acceptable hemp THC level, and includes the testing results available upon request by DSHS before distribution or sale. See id. at § 300.303(k).
DSHS shall conduct randomized testing of consumable hemp products at Texas retail stores and other facilities engaged in the manufacturing, processing, or distributing of consumable hemp products to ensure these products fall within the acceptable THC level, are in compliance with the USDA’s Hemp Production Plan, and do not contain any harmful ingredients. See id. at § 300.302(c). Additionally, according to the DSHS Proposed Rules, it is mandatory that processors and manufacturers conduct testing and sampling “using acceptance criteria that are protective of public health.” Id. at § 300.303(j). For purposes of clarity, this seemingly vague provision should be addressed to DSHS via comments and concerns during the 31-day public comment window.
Furthermore, DSHS may request that samples of representative raw or finished consumable hemp products be sent to the department at the expense of the consumable hemp products license holder, owner, or registrant. See id. at § 300.302(d)-(e). Under the DSHS Proposed Rules, a “registrant” is a person who “sells consumable hemp products directly to consumers, and who submits a complete registration form to the department for purposes of registering their place of business to sell consumable hemp products at retail to the public.” Id. at § 300.101(33). Essentially, a “registrant” is a properly registered retailer of consumable hemp products.
Additional Provisions Related to Testing
Upon request by DSHS, hemp distributors, manufacturers, and processors, must provide the test results of their consumable hemp products to the department. See id. at § 300.303(b). Registrants must provide their test results to DSHS or to consumers upon request. See id. at § 300.303(c). Consumable hemp products that are adulterated in any manner or that exceed the acceptable hemp THC level must not be sold at retail or otherwise introduced into commerce in Texas. See id. at § 300.303(a).
Under the DSHS Proposed Rules, consumable hemp products license holders must use independent testing accredited laboratories where they hold no ownership interest in the accredited laboratory or, if the accredited laboratory is a publicly-traded company, where they hold less than a ten percent interest in the accredited laboratory. See id. at § 300.303(d). License holders must pay all costs of raw and finished hemp product testing as determined by the accredited laboratory they select. See id. at § 300.303(e).
DSHS must recognize and accept an accredited laboratory’s test results, DSHS may require that a copy of such test results be sent directly to the department and the consumable hemp products license holder, and DSHS shall notify the license holder of the test results within 14 days after the department received them. See id. at § 300.303(f)-(h). License holders must retain test results from samples for at least three years from the date these results are made available to them. See id. at § 300.303(i).
Preview of Parts IV-VI:
In Parts IV through VI of this Series, I will analyze the provisions of the DSHS Proposed Rules regarding registration for retailers of consumable hemp products and the regulations on the retail sale of consumable hemp products. Lastly, I will explore violations under the DSHS Proposed Rules and how DSHS enforces its various rules and regulations concerning consumable hemp products. Stay tuned as we conclude with our hemp lawyers identifying their top issues with the DSHS Proposed Rules.
Paul Stevenson is an associate attorney with Ritter Spencer Cheng PLLC based in the Dallas office, who represents all sectors of the hemp industry, including manufacturers, distributors, and retailers of consumable hemp products. As cannabis and hemp lawyers, the lawyers at Ritter Spencer Cheng are prepared to advise your hemp or cannabis business in all facets of the industry, including formation and transactional issues and in cannabis litigation. Contact Ritter Spencer Cheng or give us a call at 214.295.5070 for more information.